


When a person tries to move their body, nerve signals are transmitted from the brain to the muscles via motor neurons, causing the musculoskeletal system, including joints, to move. Just before the body movement, faint "bio-electrical signals" that reflect a person's intention leak out to the skin surface. HAL can read this signal and control the power unit to assist joint movement in unison with the person. By using HAL, which utilizes the bio-electrical signal that reflects the wearers’ intended motion, interactive biofeedback is promoted in and outside of the body between HAL, brain, nervous system, and muscular system. Cybernics Treatment is based on this iBF theory (interactive bio-feedback theory). The effect of this treatment method to promote functions of the lower limb, upper limb, and body trunk is being reported through clinical application. In other words, the key to promoting functional improvement through Cybernics Treatment is not just transmitting voluntary command signals from the human brain to the musculoskeletal system via the spinal cord and peripheral nerves, but more so on the return of feedback from the sensation of actual movement to the human brain. The functional improvement/regeneration technology using HAL based on this iBF Theory is an innovative treatment technology that enables improvement/regeneration of human brain-nerve-musculoskeletal functions by activating the body's self-healing ability that humans are naturally in possession of.

CYBERDYNE has established Cyberdyne Care Robotics GmbH in Bochum, NRW, Germany, with BG RCI (Public Workers' Compensation Insurance Institution) as a business partner to provide Cybernics Treatment for patients with spinal cord injury, stroke, and many more. Cyberdyne Care Robotics GmbH was established in Bochum, Germany. This was a major step toward making Cybernics Treatment a global standard treatment. In 2013, Europe's largest third-party certification body certified conformity of HAL to the European Medical Device Directive (MDD). With this certification, the HAL will be affixed with the CE marking for medical device classification, which requires periodic review. It can now be distributed and sold as a medical device throughout the EU. It is expected to be deployed as a function-improving treatment for diseases and injury in preparation for various insurance coverages throughout the EU. The DGUV (German Public Accident Insurance) workers' compensation insurance covers the entire 500 euros (about 65,000 yen) per treatment x 60 sessions.
In cooperation with NEDO (New Energy and Industrial Technology Development Organization), the State of NRW, BG Bergmanns Heil Labor Hospital, and University of Tsukuba, the Company schedules HAL's demonstration tests and promotes its implementation in society.
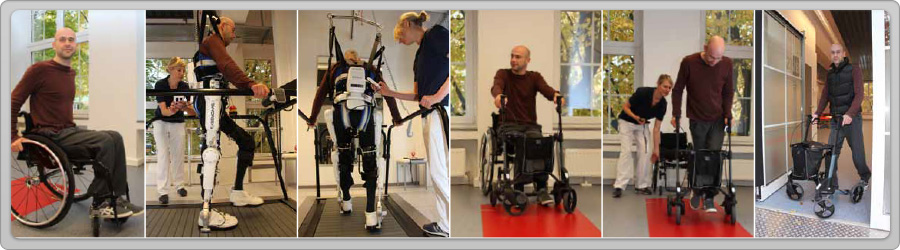
Philippe von Gliszynski was paralyzed below the 12th thoracic vertebrae in 2010 when he fell 3 meters down from the roof while clearing snow. He had only a slight feeling in his left leg. After one surgery and the conventional rehabilitation measures, he had to be prepared to live in a wheelchair for the rest of his life. However, in February 2012, when he participated in a clinical trial of HAL. After 3 months in June 2012, he was able to walk 10 meters in only 26 seconds, compared to 72 seconds with a walker at the beginning of the sessions. With subsequent practice, he was able to walk with a walker more than 1,000 meters after taking off HAL. This was unimaginable at the beginning of the treatment.

© 2026 CYBERDYNE INC. ALL RIGHTS RESERVED.